男女共同参画学協会連絡会
支援企業による広告記事
- ソーラボジャパン株式会社
- CAD を使って光学装置を設計してみよう
- 「生物物理」2025年10月号
- ソーラボジャパン株式会社
- 次世代2光子顕微鏡―小型化がもたらす新たな可能性
- 「生物物理」2024年10月号
- ソーラボジャパン株式会社
- サイエンティフィックカメラと周辺機器の同期
- 「生物物理」2023年10月号
- ソーラボジャパン株式会社
- 顕微鏡のリノベーション ~ 顕微鏡ポートを活用した光学系の導入
- 「生物物理」2022年12月号
「Biophysics and Physicobiology」に Hironori Hayashi, Miyuki Kato-Murayama, Takeshi Kurasawa, Eiichi N. Kodama, Mikako Shirouzu, Kazuhiko Igarashi, Kazutaka Murayama による "Liquid-liquid phase separation and its regulation of the BACH2 intrinsically disordered region" をJ-STAGEの早期公開版として掲載
2026年02月25日 学会誌
日本生物物理学会欧文誌[Biophysics and Physicobiology]に以下の論文が早期公開されました。
Hironori Hayashi, Miyuki Kato-Murayama, Takeshi Kurasawa, Eiichi N. Kodama, Mikako Shirouzu, Kazuhiko Igarashi, Kazutaka Murayama
"Liquid-liquid phase separation and its regulation of the BACH2 intrinsically disordered region"
URL:https://doi.org/10.2142/biophysico.bppb-v23.0010
- Abstract
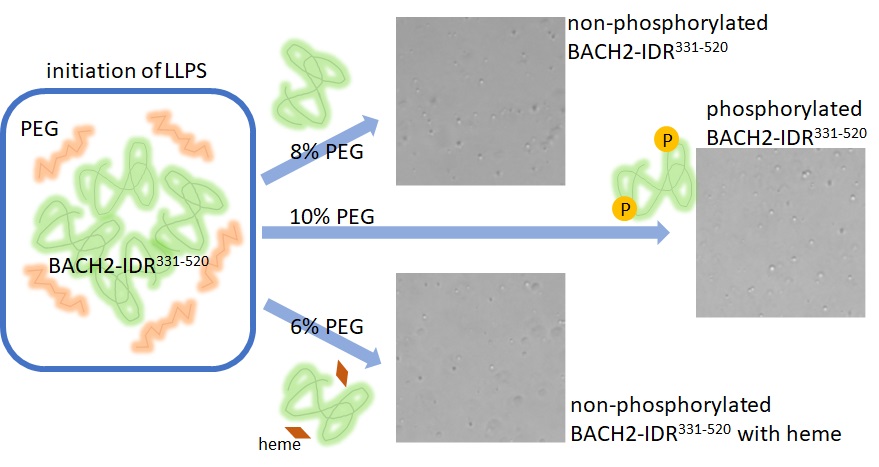
- Intrinsically disordered regions (IDRs) are vital for several cellular processes. They play a significant role in liquid-liquid phase separation (LLPS). LLPS enhances reaction efficiency by locally concentrating proteins and nucleic acids. The BACH2 transcription factor contains such IDRs. Early observations hinted at the phase separation capabilities of BACH2. However, how BACH2-IDR directly participates in LLPS is not fully understood. Its precise modulation by heme binding and phosphorylation also requires further elucidation. This study experimentally demonstrated the intrinsic capability of BACH2-IDR (residues 331-520) to undergo LLPS in vitro. Microscopic observations revealed that TANK-binding kinase 1(TBK1)-mediated phosphorylation suppressed LLPS of BACH2-IDR. Conversely, the coexistence of heme enhanced LLPS, initiating LLPS at lower polyethylene glycol concentrations. Bioinformatic analyses supported these experimental observations. Tools such as FuzDrop and CIDER were used to predict LLPS propensity from sequences. Specifically, the calculations for the sequence of phosphorylation-mimic mutations showed changes in LLPS-promoting regions. Heme binding influenced TBK1-mediated phosphorylation sites. Some of these sites overlap with predicted LLPS-driving regions. Heme binding also induced substantial conformational alterations within BACH2-IDR. Consequently, we propose LLPS as a fundamental regulatory mechanism for BACH2 protein function. This process complements its known regulation through heme binding and phosphorylation.
URL: https://doi.org/10.2142/biophysico.bppb-v23.0010