1
An article with the title "Investigation on substrate specificity and catalytic activity of serine protease neuropsin" by Masami Lintuluoto, Mitsumasa Abe, Yota Horioka, Yoshifumi Fukunishi, Hideki Tamura, Juha M. Lintuluoto is published as the J-STAGE Advance Publication.
2022 September 22 BPPB
A following article is published as the J-STAGE Advance Publication in "Biophysics and Physicobiology".
Masami Lintuluoto, Mitsumasa Abe, Yota Horioka, Yoshifumi Fukunishi, Hideki Tamura, Juha M. Lintuluoto
"Investigation on substrate specificity and catalytic activity of serine protease neuropsin"
URL:https://doi.org/10.2142/biophysico.bppb-v19.0040
- Abstract
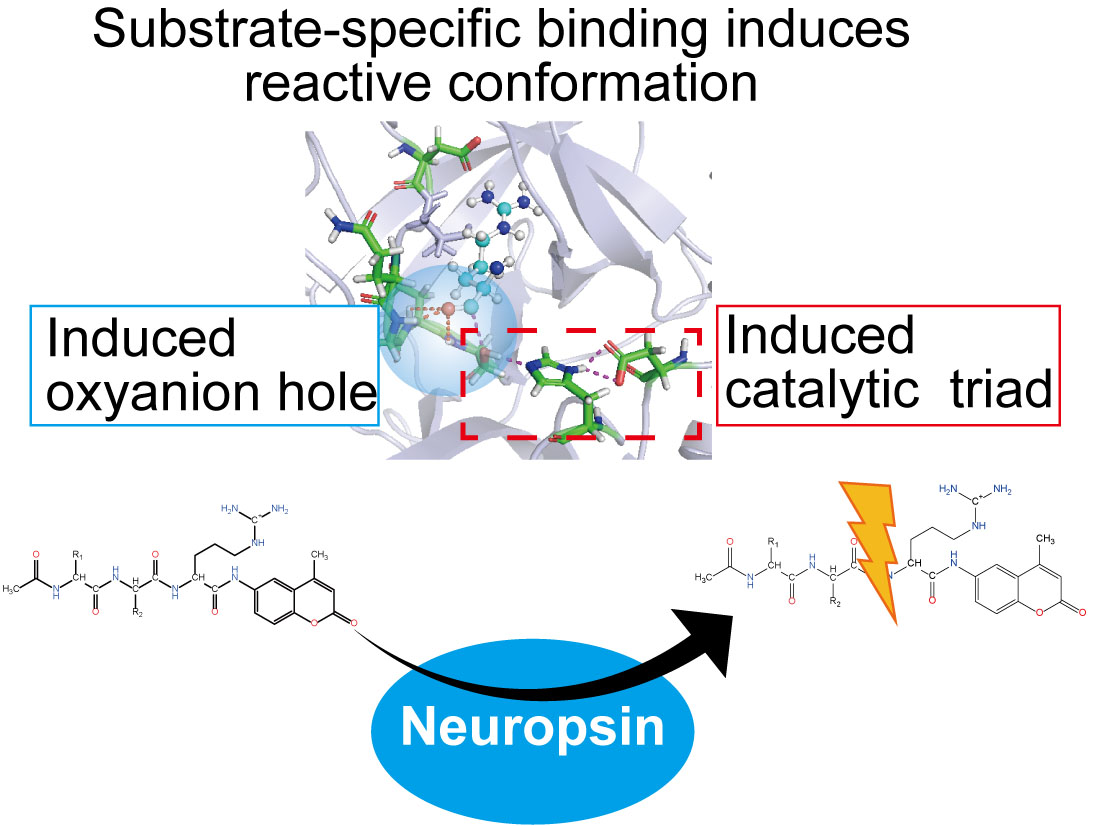
- Neuropsin is one of serine proteases mainly found at the hippocampus and the amygdala, where it contributes to the long-term potentiation and memory acquisition by rebuilding of synaptic connections. Despite of the importance of neuropsin, the substrate specificity and regulation mechanisms of neuropsin have been unclear. Thus, we investigated the substrate specificity and the catalytic activity of neuropsin by the protein-ligand docking and molecular dynamics (MD) simulations and succeeded to reproduce the trend of the experimental results. Our study revealed that the substrate specificity and the activity of neuropsin depended on multiple factors: the substrate charge, the substrate orientation, the hydrogen bond network within the catalytic triad and the substrate, and the formation of the oxyanion hole. The apo neuropsin was not reactive without proper alignment of catalytic triad. The substrate binding induced the reactive alignment of catalytic triad. Then the substrate-neuropsin interaction forms the oxyanion hole that stabilizes the transition state and reduces the free-energy barrier of the following scission reaction.